Introduction
Have you ever passed early stability testing, only to see moisture-related issues appear months later – when packaging changes are no longer an option?
In pharmaceutical packaging, these failures rarely occur because desiccants are missing. More often, the issue is that the desiccant system was never engineered for the product. Bottles and blisters allow moisture ingress over time, and when adsorption behavior, response speed, or capacity are mismatched, degradation begins quietly and progressively.
This guide is grounded in real packaging projects. It explains how pharmaceutical desiccants perform in practice, how to size them correctly, and how to align material selection with stability and regulatory requirements, so moisture becomes a controlled design variable, not a late-stage surprise.
Not All Pharmaceutical Desiccants Behave the Same
Desiccants differ not only in chemical composition, but also in consistency, adsorption kinetics, and suitability for specific formulations.
Material | Type | Key Characteristic | Typical Applications |
Silica Gel | Adsorbent | Stable, linear adsorption, excellent batch consistency | Tablets, capsules, diagnostics |
Molecular Sieve | Adsorbent | Extremely aggressive at low RH (<10%) | Effervescent tablets, lyophilized drugs |
Montmorillonite Clay | Adsorbent | Natural, economical, regenerable | Nutraceuticals, standard oral solids |
Activated Carbon | Adsorbent | VOC and odor adsorption | Odor‑sensitive formulations |
Below is how these materials behave in practical pharmaceutical packaging scenarios.
Silica Gel
Silica gel remains the most widely used pharmaceutical desiccant. Its primary advantage lies in its linear adsorption profile, as relative humidity increases, moisture uptake rises steadily and predictably.
Because it does not aggressively remove moisture at low RH, silica gel helps avoid over-drying issues such as brittle capsule shells, tablet cracking, or excessive static buildup. This makes it well suited for formulations that require controlled humidity rather than rapid moisture removal.
Molecular Sieves
When moisture sensitivity is critical, molecular sieves are often the only viable option.
Their uniform pore structure (typically 3Å or 4Å) allows them to capture water molecules extremely quickly. Unlike silica gel, molecular sieves maintain strong adsorption capacity even at very low relative humidity and elevated temperatures.
Montmorillonite Clay
Montmorillonite clay is a naturally occurring calcium aluminosilicate and remains a cost-effective solution for many solid oral dosage forms.
It performs well under standard temperature and humidity conditions and is widely used in both food and pharmaceutical applications. While its adsorption capacity decreases at sustained temperatures above 120°F (49°C), it offers strong advantages for brands focused on sustainability and “natural” positioning.
Activated Carbon
Activated carbon is not primarily a desiccant, but it plays a critical supporting role.
Its extremely high surface area allows it to adsorb odors and volatile organic compounds (VOCs). In pharmaceutical packaging, it is often combined with silica gel or clay when odor control impacts patient compliance.
How to Select the Right Desiccant for Your Formulation
Selecting a desiccant is not about choosing a packet size, it is about matching material behavior to drug stability requirements.
Understanding Adsorption Isotherms
An adsorption isotherm describes how much moisture a desiccant holds at a given temperature as relative humidity increases.
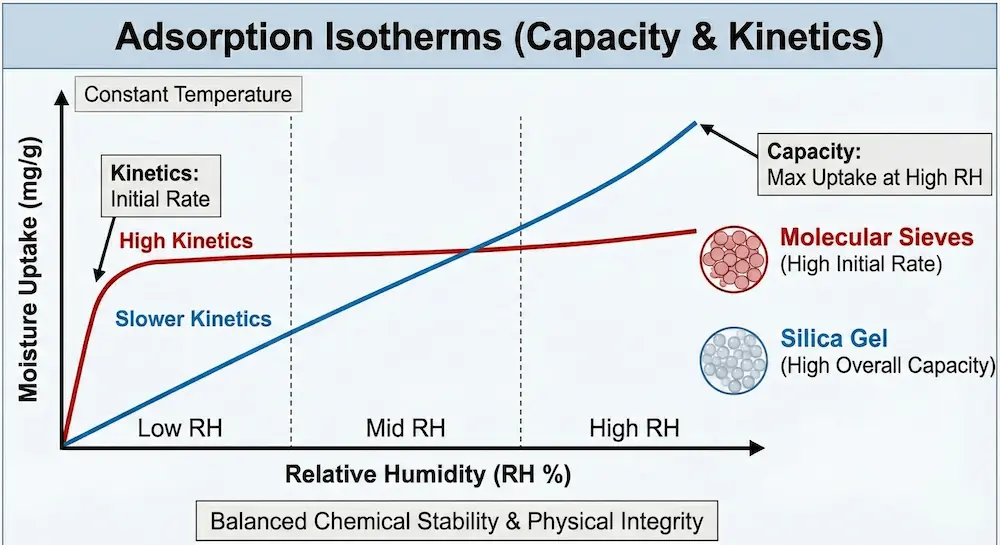
- Silica gel shows a gradual, linear uptake, making it suitable when controlled drying is required.
- Molecular sieves exhibit a steep initial curve, rapidly removing moisture from the headspace.
Understanding this behavior is essential when balancing chemical stability against physical integrity.
Reaction Kinetics and Target ERH
Defining the required equilibrium relative humidity is essential.
If a drug degrades via hydrolysis at 30% RH, a desiccant system that stabilizes at 40% RH offers no real protection. At the same time, removing too much moisture can damage gelatin capsules or alter tablet hardness.
In practice:
- Use molecular sieves when reaction kinetics demand immediate moisture removal
- Use silica gel or clay when maintaining a controlled, moderate humidity is more important than speed
Calculating the Required Quantity
A simplified engineering approach looks like this:
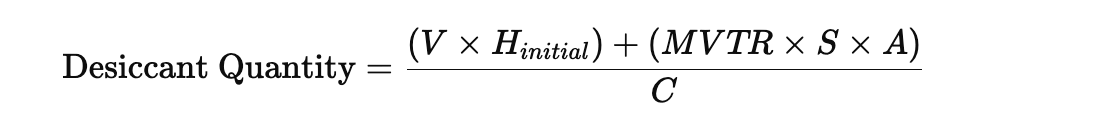
Where:
- V = Headspace volume
- H₍initial₎ = Initial humidity at sealing
- MVTR = Moisture Vapor Transmission Rate of the package
- S = Shelf life (days)
- A = Package surface area
- C = Desiccant capacity per unit
While general rules of thumb exist, accurate sizing almost always requires calculation, especially for long shelf lives or moisture-sensitive formulations.
Delivery Formats and Packaging Integration
Performance alone is not sufficient, the desiccant must also integrate smoothly into your packaging line and patient experience.
Drop-in Formats
- Canisters: Ideal for high-speed, automated bottling lines. Their rigid geometry minimizes feeding issues.
- Sachets/Packets: Typically made from medical-grade Tyvek or coated paper. Tyvek offers excellent strength and breathability, although sachets may slow insertion at very high line speeds.
Integrated Solutions
Packaging trends increasingly favor built-in moisture control:
- Desiccant closures and liners reduce component count and free up internal volume
- Desiccant blister films protect each individual dose
An important safety benefit of integrated solutions is the elimination of loose components, reducing ingestion risk, especially for pediatric and geriatric products.
Regulatory Standards (FDA, USP, ISO)
In pharmaceutical packaging, documentation matters as much as performance.
A qualified desiccant supplier should be able to support:
- USP <670> compliance for pharmaceutical desiccants
- FDA 21 CFR food and drug contact safety
- An active Type III Drug Master File (DMF) for regulatory cross-referencing
- ISO 15378 certification confirming GMP manufacturing conditions
Without this foundation, even technically sound solutions can become regulatory obstacles.
Emerging Trends in Active Packaging
Sustainability and Natural Materials
As ESG commitments increase, interest in bio-based plastics and naturally occurring minerals continues to grow. Montmorillonite clay, already well-established in food applications, is gaining wider adoption in pharmaceutical packaging due to its regenerability and lower carbon footprint.
Intelligent Packaging
Next-generation systems go beyond absorption. Smart desiccants and moisture-sensing packaging are being developed to record humidity exposure throughout the supply chain, providing visibility that was previously unavailable for sensitive drugs and biologics.
FAQs on Pharmaceutical Desiccant
What is the difference between silica gel and molecular sieves in pharmaceuticals?
Molecular sieves remove moisture faster and to much lower RH levels. Silica gel provides more controlled, gradual moisture management.
Can desiccant canisters be used with liquid medications?
No. Desiccants are designed exclusively for solid dosage forms such as tablets, capsules, and powders.
Is Montmorillonite clay safe for oral products?
Yes. It complies with USP <670> and is widely used for oral solid dosage forms.
How do you calculate the required desiccant unit for a 100cc bottle?
There is no universal answer. While 1 gram is often used as a starting point, accurate sizing requires MVTR and headspace calculations.
Ready to Optimize Your Stability Profile?
Shelf life should never be left to assumption.
Whether your product requires aggressive moisture control or a sustainable, natural solution, accurate selection and calculation make the difference between stability and failure.
Request a Technical Consultation to review your packaging system and adsorption requirements
Reliable moisture control starts with informed design.