It is tough to control humidity inside sealed pharmaceutical containers.
Just a small amount of trapped moisture can ruin a whole batch of solid-dose medications. Moisture causes hydrolysis and oxidation, which eat away at your active pharmaceutical ingredients (APIs). A few weeks of fluctuating warehouse temperatures, and you could be looking at degraded medicine, compromised dissolution rates, and costly compliance headaches.
Here is a practical, technical guide to using medical desiccant silica gel. We’ll show you how to properly integrate these desiccants into your packaging line so your products stay safe and stable.
How Medical Silica Gel Works
Typically, silica gel traps water vapor within its internal structure, maintaining a dry environment inside your packaging. It acts like a physical sponge, not a chemical one.
Medical-grade silica gel (silicon dioxide, SiO₂) is completely non-corrosive and chemically inert. This means it won’t react with your APIs or excipients. Instead, it relies on a massive network of microscopic pores to physically pull moisture from the air.
Setting Your Moisture Control Targets
When you package solid oral dosage forms, your main goal is to keep the headspace humidity below the drug’s critical relative humidity (CRH).
- Target Humidity: You generally want to reduce headspace relative humidity to below 20%. If your formulation is highly hygroscopic, you might need to push that below 10%.
- Adsorption Capacity: High-quality silica gel is powerful. It can adsorb 30% to 40% of its weight in water vapor when fully saturated.
By buffering these humidity swings, silica gel prevents moisture from migrating into your capsules. It also stops microbial growth dead in its tracks, because microbes need high water activity to survive.
Meeting Regulatory Compliance and Standards
In the pharmaceutical world, a desiccant is a “contact material.” That means it has to meet the exact same rigorous safety standards as the pill bottle itself.
If you’re sourcing medical desiccants, you need to verify a few strict compliance metrics.
USP Auxiliary Packaging Components
The United States Pharmacopeia (USP) sets the rules for desiccants under General Chapter <670>. You must ensure your desiccants pass these tests:
- Adsorption Capacity: This proves the desiccant actually absorbs moisture as claimed at specific humidity levels like 40% and 80%.
- Visual Inspection: This guarantees there is no foreign matter or weird discoloration in your packets.
- Soluble Ionizable Substances: This limits any leachable ions that might interact with your drugs.
FDA 21 CFR Compliance
Your materials must also align with FDA regulations for indirect food and drug contact.
- Silica Gel Core: The silica beads themselves must be Generally Recognized As Safe (GRAS). You can read more about this in 21 CFR 182.1711.
- Sachet Material: The pouch holding the gel (usually Tyvek®) has to comply with rules regarding olefin polymers. This ensures the packaging doesn’t leach plastics into your medicine.
- Drug Master File (DMF): We highly recommend working with suppliers who hold an active Type III DMF with the FDA. This lets you reference their manufacturing data in your own FDA filings without headaches.
Indicator Safety
Older moisture indicators used cobalt chloride, which turns from blue to pink. However, the EU now classifies this as a carcinogen.
If you need indicators, you should exclusively use cobalt-free versions. Modern indicators use safe iron salts that shift from orange to green, keeping you compliant with global safety directives.
Choosing the Right Packaging Format
The shape of your desiccant depends entirely on your packaging line’s automation setup and the size of your primary container.
Desiccant Canisters
Rigid cylindrical canisters are usually the best choice for high-speed automated bottling lines.
- Why they work: Their hard, uniform shape prevents them from jamming in hopper-fed insertion machines. You can run lines at over 300 bottles per minute without worrying about dusting or packet tears.
Desiccant Packets and Sachets
Flexible sachets give you a much lower profile and a higher surface-area-to-volume ratio.
- Why they work: They flatten out easily and fit into tight blister packs or diagnostic kits. They also typically cost less per unit than rigid canisters. For safe materials, we recommend Tyvek because it won’t burst under pressure.
Continuous Strip Desiccants
These are desiccant packets connected together on a long reel.
- Why they work: If you use specialized dispensing machines, these reels feed directly into the cutter. This cuts down on the downtime you’d normally spend refilling loose hoppers.
You can explore our full lineup of pharmaceutical packaging desiccants to see which format fits your specific machinery.
How to Calculate Your Desiccant Needs
You shouldn’t just drop an arbitrary 1-gram packet into every bottle. Guessing your desiccant weight is a huge regulatory risk.
To get it right, you need to calculate your required dosage by evaluating three sources of moisture: the air trapped in the headspace, the initial moisture of the pills, and the moisture that will permeate the bottle over time.
The Basic Sizing Formula
Engineers use a specific calculation to find the exact weight of desiccant required. Here is the formula:
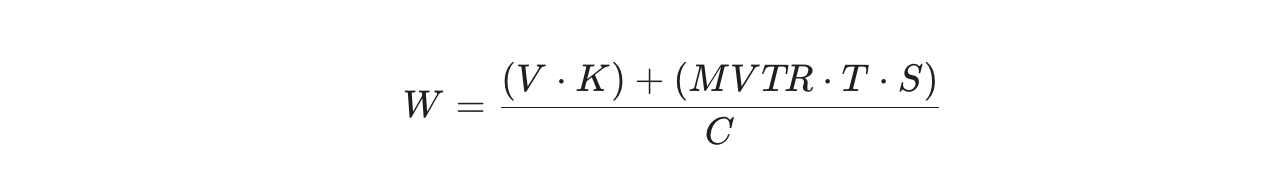
Where:
- W = Weight of desiccant required (grams)
- V = Volume of the headspace
- K = Moisture factor of the trapped air
- MVTR = Moisture Vapor Transmission Rate of your container
- T = Desired shelf life in days
- S = Your safety factor
- C = The adsorption capacity of the desiccant
Testing and Validation
Math gives you a great starting point, but you still have to prove it in the lab. Accelerated stability testing is mandatory.
You will typically test your packaging at 40°C and 75% relative humidity for 6 months. If your drug’s moisture content spikes during this test, you might need to increase your desiccant mass or buy a thicker bottle.
Securing Your Supply Chain
The global packaging market changes constantly. If you want a reliable supply of medical-grade silica gel, you need to vet your vendors carefully.
Always look for manufacturing facilities that hold ISO 15378 certifications. This proves they follow Good Manufacturing Practices (GMP) for primary packaging. You should also demand a Certificate of Analysis (CoA) for every single batch so you can trace the raw materials.
Taking the time to do this right ensures your products retain their full potency from the manufacturing floor all the way to the patient’s hands.
Conclusion
You now know exactly why medical-grade silica gel is a must-have for your packaging line.
But figuring out the exact gram weight to pass your accelerated stability tests can still be a headache. You don’t have to guess or risk failing your validation protocols.
We want to hear from you. Which packaging format are you currently working on?
- Are you running a high-speed bottling line that needs rigid canisters?
- Or are you designing tight blister packs that require low-profile Tyvek sachets?
Let us do the heavy lifting for your compliance. Send us your container volume, target shelf life, and packaging material. We’ll run the MVTR calculations and tell you exactly which desiccant size you need to protect your specific drug formulation.
Click here to get your free desiccant calculation or reach out to our engineering team today!




